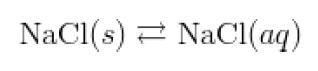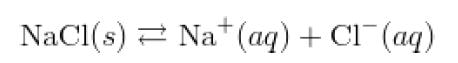Scientific Method is a series of organized steps that are taken to answer a question or solve a problem.
Example:
1. You noticed that ice melted fast in water.
2. Does ice melt faster in different liquid (salt solution)?
3. Ice will melt faster in salt solution than it will in water.
4. Set up an experiment with a glass of salt solution, a glass of water and an ice cube for each.
5. Record what is happening as well as the results. When each ice cube completely melted.
6. Ice does not melt faster in different liquid (salt solution).
Example:
1. You noticed that ice melted fast in water.
2. Does ice melt faster in different liquid (salt solution)?
3. Ice will melt faster in salt solution than it will in water.
4. Set up an experiment with a glass of salt solution, a glass of water and an ice cube for each.
5. Record what is happening as well as the results. When each ice cube completely melted.
6. Ice does not melt faster in different liquid (salt solution).
Quarter 1 – Module 7: Concentration of Solutions
Quarter 1 – Module 6: Solutions
What I Need to Know
Solutions are substances that are very familiar to us. The air we breathe is a solution of several gases. The sea water we enjoy to swim is a solution of water and various minerals from the earth’s crust. Most of the jewelry we wear are solutions of precious metals in gold or silver. Coins are solutions of copper, silver and nickel. Most of the food we eat and the juice we drink are solutions of different kind of substances. To better understand what solutions are, we must first know how they are formed, the types in which they come, the factors that affect their formation and the rate affecting their solubility.
After going through this module, you are expected to:
1. investigate properties of saturated or unsaturated solutions.( S7MT-Ic-2);
2. explain different factors affecting solubility and
3. show self -reliance in performing the task at home independently.
Solutions are substances that are very familiar to us. The air we breathe is a solution of several gases. The sea water we enjoy to swim is a solution of water and various minerals from the earth’s crust. Most of the jewelry we wear are solutions of precious metals in gold or silver. Coins are solutions of copper, silver and nickel. Most of the food we eat and the juice we drink are solutions of different kind of substances. To better understand what solutions are, we must first know how they are formed, the types in which they come, the factors that affect their formation and the rate affecting their solubility.
After going through this module, you are expected to:
1. investigate properties of saturated or unsaturated solutions.( S7MT-Ic-2);
2. explain different factors affecting solubility and
3. show self -reliance in performing the task at home independently.
What I Know
Directions: Read carefully each item. Use a separate answer sheet for your answers. Shade only the letter of the best answer for each question.
1. Which of the following is NOT a solution?
A. Amalgam
B. Mayonnaise
C. Oxygen dissolved in water
D. Sugar dissolved in water
2. Which is an example of a solution?
A. Blood
B. Cooked flour
C. Marshmallow
D. Seawater
3. Which of the following statements is correct?
A. Pressure changes have little effect on solubility if the solute is a liquid or solid.
B. All solids are soluble in a liquid as temperature rises
C. A supersaturated solution is an example of a dynamic system.
D. The solubility of a gas in a liquid usually increases as temperature increases.
4. Decreases in vapour pressure will cause________________.
A. An increase in boiling point
B. A decrease in boiling point
C. No effect
5. A solubility of CO2 gas in liquid will decrease if
A. Pressure is increased
B. Temperature is decreased
C. Temperature is increased
6. The two components of a solution are solute and solvent. Which statement
describes the solute?
A. It is the liquid form in the solution
B. It is the liquid component of a solution
C. It is the component of solution in smaller quantity.
D. It is the component of solution in bigger quantity.
7. The two components of a solution are solute and solvent. Which statement
describes the solvent?
A. It is the liquid form in the solution
B. It is the liquid component of a solution
C. It is the component of solution in smaller quantity.
D. It is the component of solution in bigger quantity.
8. Which of the following statements BEST describes a homogeneous solution?
A. It is usually liquid.
B. It contains a solute and solvent.
C. It can be dilute or concentrated
D. Its components are distributed evenly in the solution
9. What opposing processes occur in a saturated solution?
A. Dissociation and combination
B. Dissolution and crystallization
C. Oxidation and reduction
D. Vaporization and condensation
10. What kind of solution forms when gasoline evaporates in air?
A. Liquid –in-liquid solution
B. Liquid –in-gas solution
C. Gas-in-gas solution
D. Gas-in-liquid solution
Directions: Read carefully each item. Use a separate answer sheet for your answers. Shade only the letter of the best answer for each question.
1. Which of the following is NOT a solution?
A. Amalgam
B. Mayonnaise
C. Oxygen dissolved in water
D. Sugar dissolved in water
2. Which is an example of a solution?
A. Blood
B. Cooked flour
C. Marshmallow
D. Seawater
3. Which of the following statements is correct?
A. Pressure changes have little effect on solubility if the solute is a liquid or solid.
B. All solids are soluble in a liquid as temperature rises
C. A supersaturated solution is an example of a dynamic system.
D. The solubility of a gas in a liquid usually increases as temperature increases.
4. Decreases in vapour pressure will cause________________.
A. An increase in boiling point
B. A decrease in boiling point
C. No effect
5. A solubility of CO2 gas in liquid will decrease if
A. Pressure is increased
B. Temperature is decreased
C. Temperature is increased
6. The two components of a solution are solute and solvent. Which statement
describes the solute?
A. It is the liquid form in the solution
B. It is the liquid component of a solution
C. It is the component of solution in smaller quantity.
D. It is the component of solution in bigger quantity.
7. The two components of a solution are solute and solvent. Which statement
describes the solvent?
A. It is the liquid form in the solution
B. It is the liquid component of a solution
C. It is the component of solution in smaller quantity.
D. It is the component of solution in bigger quantity.
8. Which of the following statements BEST describes a homogeneous solution?
A. It is usually liquid.
B. It contains a solute and solvent.
C. It can be dilute or concentrated
D. Its components are distributed evenly in the solution
9. What opposing processes occur in a saturated solution?
A. Dissociation and combination
B. Dissolution and crystallization
C. Oxidation and reduction
D. Vaporization and condensation
10. What kind of solution forms when gasoline evaporates in air?
A. Liquid –in-liquid solution
B. Liquid –in-gas solution
C. Gas-in-gas solution
D. Gas-in-liquid solution
What is It
Classification of Solutions According to their Phase
If solutions are classified according to their phase, there are three types of solutions. These are the solid solution, the liquid solution, and the gaseous solution. The illustration below presents the three types of solutions based on their phase.
Classification of Solutions According to their Phase
If solutions are classified according to their phase, there are three types of solutions. These are the solid solution, the liquid solution, and the gaseous solution. The illustration below presents the three types of solutions based on their phase.
Gaseous Solution - includes gases or vapors dissolved in one another. Two or more gases can form a solution. Air is an example of gaseous solution. When dry is made up of oxygen gas dissolved in nitrogen gas.
Liquid Solution – contain a liquid solvent in which gas, liquid, or solid is dissolved. Water is the most common liquid solution. Many things can be dissolved in it. Table salt is an example of a solid dissolved in liquid. A liquid and a gas can also be dissolved in a liquid solution.
Solid solution – is a mixture of solids spread equally throughout one another. Metal is an example of a solid solution at room temperature.
Saturation of Solution
The ratio of the amounts of solutes and solvents may also be used to classify solutions. A solution which contains, at a specific temperature, an amount of solute that it can normally hold in the presence of the given amount of solvent is a saturated solution. A solution is unsaturated if it contains less solute than what it can normally hold. A solution is supersaturated if it contains more solute than what it can normally hold.
For instance, 100g of water can dissolve no more than 40g of sugar at 250C. If we have 100g of water at 250C, and we add 40g of sugar to produce a solution, the solution form is saturated. If we add 35g of sugar (or any amount that is less than 40g), the solution produced is unsaturated. However, if solution holds 45g of dissolved sugar (or any amount that is more than 40g) the solution is super saturated.
At the laboratory, the term “dilute” and “concentrated” are usually used to express the concentration of a given solution. Dilute solution are those that contain a small quantity of solute relative to the amount of solvent present. Concentrated solutions are those that contain large amounts of solute compared to the amount of
solvent present.
It should be noted, however, that using the term saturated, unsaturated, supersaturated, concentrated or dilute is not encouraged, if the exact concentration of the solution is desired. More accurate methods of expressing the concentrations of solution such as percentage by mass and percentage by volume are used. These method will be discussed in a separate module.
Saturated and Unsaturated Solutions
Table salt (NaCl) readily dissolves in water. Suppose that you have a beaker of water to which you add some salt, stirring until it dissolves. So you add more and that dissolves. You keep adding more and more salt, eventually reaching a point that
no more of the salt will dissolve no matter how long or how vigorously you stir it.
Why? On the molecular level, we know that action of the water causes the individual
ions to break apart from the salt crystal and enter the solution, where they remain
hydrated by water molecules. What also happens is that some of the dissolved ions
collide back again with the crystal and remain there. Recrystallization is the
process of dissolved solute returning to the solid state. At some point the rate at
which the solid salt is dissolving becomes equal to the rate at which the dissolved
solute is recrystallizing. When that point is reached, the total amount of dissolved salt
remains unchanged. Solution equilibrium is the physical state described by the
opposing processes of dissolution and recrystallization occurring at the same rate.
The solution equilibrium for the dissolving of sodium chloride can be represented by
one of two equations.
Liquid Solution – contain a liquid solvent in which gas, liquid, or solid is dissolved. Water is the most common liquid solution. Many things can be dissolved in it. Table salt is an example of a solid dissolved in liquid. A liquid and a gas can also be dissolved in a liquid solution.
Solid solution – is a mixture of solids spread equally throughout one another. Metal is an example of a solid solution at room temperature.
Saturation of Solution
The ratio of the amounts of solutes and solvents may also be used to classify solutions. A solution which contains, at a specific temperature, an amount of solute that it can normally hold in the presence of the given amount of solvent is a saturated solution. A solution is unsaturated if it contains less solute than what it can normally hold. A solution is supersaturated if it contains more solute than what it can normally hold.
For instance, 100g of water can dissolve no more than 40g of sugar at 250C. If we have 100g of water at 250C, and we add 40g of sugar to produce a solution, the solution form is saturated. If we add 35g of sugar (or any amount that is less than 40g), the solution produced is unsaturated. However, if solution holds 45g of dissolved sugar (or any amount that is more than 40g) the solution is super saturated.
At the laboratory, the term “dilute” and “concentrated” are usually used to express the concentration of a given solution. Dilute solution are those that contain a small quantity of solute relative to the amount of solvent present. Concentrated solutions are those that contain large amounts of solute compared to the amount of
solvent present.
It should be noted, however, that using the term saturated, unsaturated, supersaturated, concentrated or dilute is not encouraged, if the exact concentration of the solution is desired. More accurate methods of expressing the concentrations of solution such as percentage by mass and percentage by volume are used. These method will be discussed in a separate module.
Saturated and Unsaturated Solutions
Table salt (NaCl) readily dissolves in water. Suppose that you have a beaker of water to which you add some salt, stirring until it dissolves. So you add more and that dissolves. You keep adding more and more salt, eventually reaching a point that
no more of the salt will dissolve no matter how long or how vigorously you stir it.
Why? On the molecular level, we know that action of the water causes the individual
ions to break apart from the salt crystal and enter the solution, where they remain
hydrated by water molecules. What also happens is that some of the dissolved ions
collide back again with the crystal and remain there. Recrystallization is the
process of dissolved solute returning to the solid state. At some point the rate at
which the solid salt is dissolving becomes equal to the rate at which the dissolved
solute is recrystallizing. When that point is reached, the total amount of dissolved salt
remains unchanged. Solution equilibrium is the physical state described by the
opposing processes of dissolution and recrystallization occurring at the same rate.
The solution equilibrium for the dissolving of sodium chloride can be represented by
one of two equations.
While this shows the change of state back and forth between solid and aqueous solution, the preferred equation also shows the dissociation that occurs as an ionic solid dissolves.
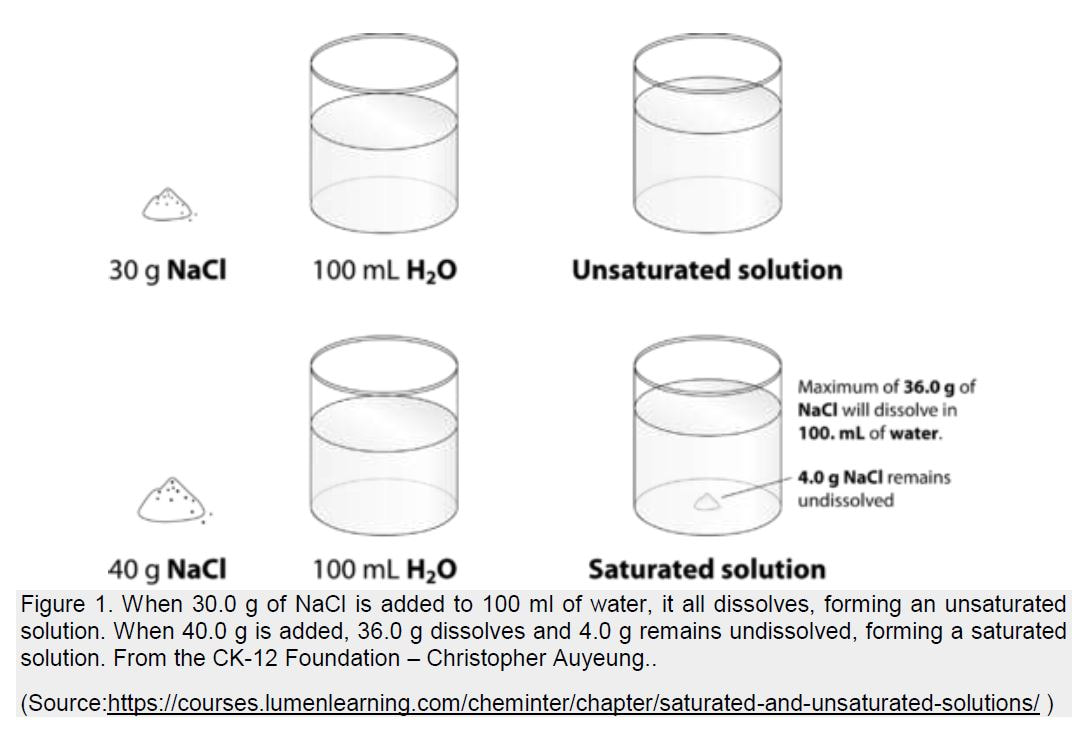
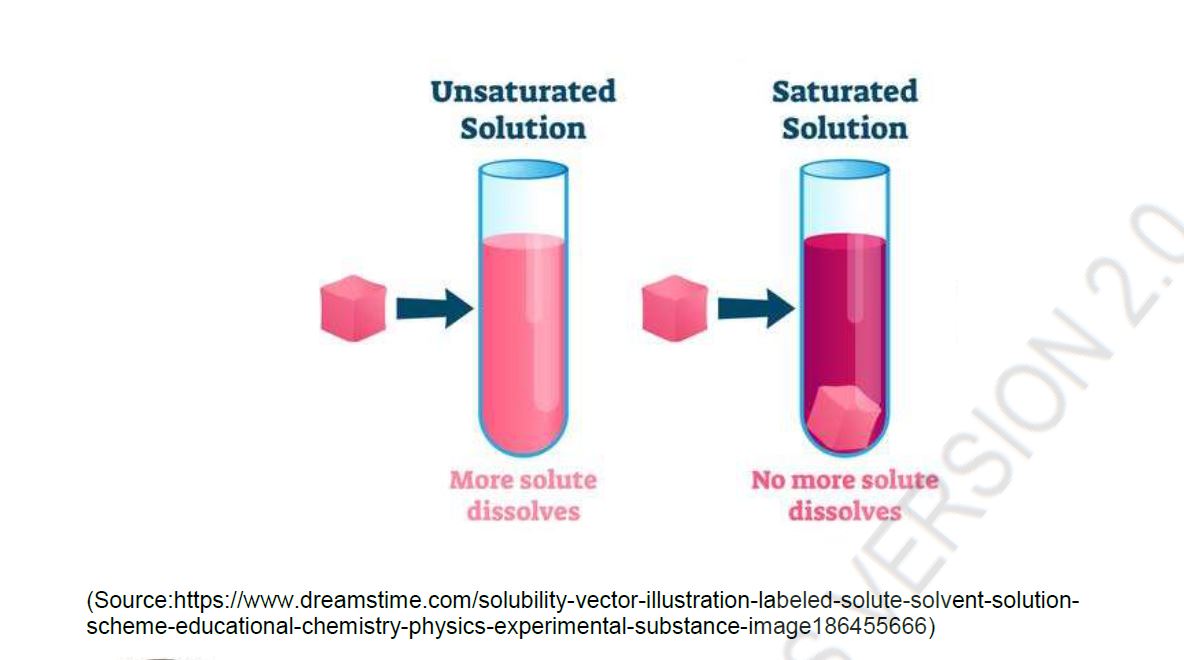
When the solution equilibrium point is reached and no more solute will dissolve, the solution is said to be saturated. A saturated solution is a solution that contains the maximum amount of solute that is capable of being dissolved. At 20°C, the maximum amount of NaCl that will dissolve in 100. g of water is 36.0 g. If any more NaCl is added past that point, it will not dissolve because the solution is saturated. What if more water is added to the solution instead? Now more NaCl would be capable of dissolving in the additional solvent. An unsaturated solution is a solution that contains less than the maximum amount of solute that is capable of being dissolved. The figure below illustrates the above process and shows the distinction between unsaturated and saturated.
How can you tell if a solution is saturated or unsaturated? If more solute is added and it does not dissolve, then the original solution was saturated. If the added solute dissolves, then the original solution was unsaturated. A solution that has been allowed to reach equilibrium but which has extra undissolved solute at the bottom of
the container must be saturated.
Factors That Affect Solubility
The extent to which the solute dissolves in a given solvent is affected by several factors like nature of the solute and solvent, temperature and pressure.
The Nature of Solutes and Solvents
The nature of solutes and solvents play the most important part in the formation of solutions, Polar solvents like water are excellent solvents for polar solutes such as sodium chloride, NaCl. Non-polar solvents like ether and benzene, have a minimal effect on the solubility of polar solutes. They are good solvents for non-polar substances such as fats and waxes. In general, polar solvents, polar solvents dissolve polar solutes and non-polar solvents dissolve non-polar solutes.
The Effects of Temperature
The effects of temperature on the solubility of solutes in solvents varies with the physical state of the solute and solvent. Most solid solutes become more soluble in liquid solvents as the temperature rises. There are only a few known solids that become less soluble in liquid solvents as temperature increases. In the solutions of gases and liquids, increase in the temperature results to decrease in the solubility of gases. This is the reason why bubbles appear when tap water is heated. Increase in temperature allows the gas molecules to escape from water molecules in the form of bubbles. The same principle is applied to soft drinks which form bubbles as they
warm up.
The Effects of Pressure
Studies reveal that pressure has very little effect on the solubility of solid solutes of liquid solvents and of solid solutes in solid solvents. However, pressure affects the solubility of gases in liquids. The amount of gas molecules that can be dissolved in a given solvent is directly proportional to the pressure. This means that if higher pressure is applied to the solution, the gas molecules are dissolved in liquids. On the other hand, less gas molecules will be dissolved in liquids at lower pressure. Note that if we open a bottle of soft drink, it “pops” and bubbles are produced. The popping sound indicates sudden change in pressure. A sealed soft drink bottle has an inside pressure of one atmosphere, and opening the bottle decreases that pressure. The solubility of the gas (carbon dioxide, CO2) molecules in liquid component of the soft drink also decreases. As a result, these gas molecules escape in the form of bubbles.
the container must be saturated.
Factors That Affect Solubility
The extent to which the solute dissolves in a given solvent is affected by several factors like nature of the solute and solvent, temperature and pressure.
The Nature of Solutes and Solvents
The nature of solutes and solvents play the most important part in the formation of solutions, Polar solvents like water are excellent solvents for polar solutes such as sodium chloride, NaCl. Non-polar solvents like ether and benzene, have a minimal effect on the solubility of polar solutes. They are good solvents for non-polar substances such as fats and waxes. In general, polar solvents, polar solvents dissolve polar solutes and non-polar solvents dissolve non-polar solutes.
The Effects of Temperature
The effects of temperature on the solubility of solutes in solvents varies with the physical state of the solute and solvent. Most solid solutes become more soluble in liquid solvents as the temperature rises. There are only a few known solids that become less soluble in liquid solvents as temperature increases. In the solutions of gases and liquids, increase in the temperature results to decrease in the solubility of gases. This is the reason why bubbles appear when tap water is heated. Increase in temperature allows the gas molecules to escape from water molecules in the form of bubbles. The same principle is applied to soft drinks which form bubbles as they
warm up.
The Effects of Pressure
Studies reveal that pressure has very little effect on the solubility of solid solutes of liquid solvents and of solid solutes in solid solvents. However, pressure affects the solubility of gases in liquids. The amount of gas molecules that can be dissolved in a given solvent is directly proportional to the pressure. This means that if higher pressure is applied to the solution, the gas molecules are dissolved in liquids. On the other hand, less gas molecules will be dissolved in liquids at lower pressure. Note that if we open a bottle of soft drink, it “pops” and bubbles are produced. The popping sound indicates sudden change in pressure. A sealed soft drink bottle has an inside pressure of one atmosphere, and opening the bottle decreases that pressure. The solubility of the gas (carbon dioxide, CO2) molecules in liquid component of the soft drink also decreases. As a result, these gas molecules escape in the form of bubbles.
Assessment
Choose the letter of the correct answer. Use the answer sheet provided.
1. What kind of solution forms when gasoline evaporates in air?
A. Liquid –in-liquid solution
B. Liquid –in-gas solution
C. Gas-in-gas solution
D. Gas-in-liquid solution
2. What opposing processes occur in a saturated solution?
A. Dissociation and combination
B. Dissolution and crystallization
C. Oxidation and reduction
D. Vaporization and condensation
3. Which of the following statements BEST describes a homogeneous solution?
A. It is usually liquid.
B. It contains a solute and solvent.
C. It can be dilute or concentrated
D. Its components are distributed evenly in the solution
4. The two components of a solution are solute and solvent. Which statement describes the solvent?
A. It is the liquid form in the solution
B. It is the liquid component of a solution
C. It is the component of solution in smaller quantity.
D. It is the component of solution in bigger quantity.
5. The two components of a solution are solute and solvent. Which statement describes the solute?
A. It is the liquid form in the solution
B. It is the liquid component of a solution
C. It is the component of solution in smaller quantity.
D. It is the component of solution in bigger quantity.
6. A solubility of CO2 gas in liquid will decrease if
A. Pressure is increased
B. Temperature is decreased
C. Temperature is increased
7. A decrease in vapour pressure will cause________________.
A. An increase in boiling point
B. A decrease in boiling point
C. No effect
8. Which of the following statements is correct?
A. Pressure changes have little effect on solubility if the solute is a liquid or solid.
B. All solids are soluble in a liquid as temperature rises
C. A supersaturated solution is an example of a dynamic system.
D. The solubility of a gas in a liquid usually increases as temperature increases.
9. Which is an example of a solution?
A. Blood
B. Cooked flour
C. Marshmallow
D. seawater
10. Which of the following is NOT a solution?
A. Amalgam
B. Mayonnaise
C. Oxygen dissolved in water
D. Sugar dissolved in water